Boron trifluoride dimethyl etherate
Boron trifluoride dimethyl etherate
CAS: 353-42-4
Molecular Formula: C2H6BF3O
Boron trifluoride dimethyl etherate - Names and Identifiers
Boron trifluoride dimethyl etherate - Physico-chemical Properties
| Molecular Formula | C2H6BF3O |
| Molar Mass | 113.87 |
| Density | 1.239g/mLat 25°C(lit.) |
| Melting Point | -14 °C |
| Boling Point | 126-127°C(lit.) |
| Flash Point | 96°F |
| Water Solubility | reacts |
| Vapor Presure | 2.99hPa at 20℃ |
| Appearance | Liquid, Fuming In Moist Air |
| Specific Gravity | 1.225 |
| Color | Clear brownish |
| BRN | 3613739 |
| Storage Condition | water-free area |
| Explosive Limit | 21.6% |
| Refractive Index | n20/D 1.308(lit.) |
| Use | Is an effective catalyst for many organic reactions, good stability, can be used as alkylation catalyst |
Boron trifluoride dimethyl etherate - Risk and Safety
| Risk Codes | R14/15 - R34 - Causes burns R48/23 - R20/22 - Harmful by inhalation and if swallowed. |
| Safety Description | S16 - Keep away from sources of ignition. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S28 - After contact with skin, wash immediately with plenty of soap-suds. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S8 - Keep container dry. S43 - In case of fire use ... (there follows the type of fire-fighting equipment to be used.) S28A - S23 - Do not breathe vapour. |
| UN IDs | UN 2965 4.3/PG 1 |
| WGK Germany | 3 |
| RTECS | ED8400000 |
| FLUKA BRAND F CODES | 10 |
| HS Code | 29319090 |
| Hazard Class | 4.3 |
| Packing Group | I |
Boron trifluoride dimethyl etherate - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| Application | Boron trifluoride-dimethyl ether complex can be used as an intermediate in pharmaceutical synthesis, such as the preparation of lithium difluorooxalate borate (LiODFB). Since LiODFB was first reported as a new type of lithium salt, due to its superior comprehensive performance, it has attracted extensive attention of researchers in the past ten years and has been reported by a large number of studies. It has been considered as the most likely new type of lithium salt to replace LiPF6. Boron methyl ether trifluoride can be used in the laboratory development process, and can also be used for the separation of boron -10 isotopes. |
| preparation | the raw material methyl ether liquid is pumped to the molecular sieve adsorption tower, and passes through the molecular sieve bed from the lower part of the molecular sieve adsorption tower to the top with a residence time of 30 minutes. The adsorption process was carried out at 0.5MpaG and 0 ℃. After molecular sieve adsorption treatment, the water content in refined methyl ether is reduced to 0.001%. The raw material boron trifluoride gas enters the low-temperature rectification tower, the top temperature of the tower is controlled at -105 ℃, and the pressure is controlled at O.1MpaG. After rectification treatment, the refined boron trifluoride comes out of the top of the tower and does not contain impurities such as hydrogen fluoride and silicon tetrafluoride. The refined methyl ether flows through the complex reactor tube from top to bottom, the refined boron trifluoride gas enters the complex reactor tube from bottom to top, and the methyl ether and boron trifluoride undergo a complex reaction on the packing of the tube. The complexation reaction is controlled at 0.6MpaG and the temperature is controlled at -10 ℃. The resulting complex is pumped to an exchange reaction distillation column. The vacuum degree of the exchange reaction distillation tower is 150mmHg, the top temperature of the tower is 85 ℃, and the temperature of the tower kettle is 90 ℃. Finally, after the exchange distillation tower reaches the exchange reaction and distillation equilibrium, boron trifluoride -11 gas with an abundance of more than 99% is obtained from the top of the tower, and boron trifluoride methyl ether -10 isotope products with an abundance of more than 95% are obtained from the tower kettle. |
| rectification process | the specific process of chemical exchange rectification of boron trifluoride-methyl ether complex is: the boron trifluoride-methyl ether complex is directly added to the rectification tower, and the distillation is heated under reduced pressure. finally, boron trifluoride -10 is concentrated in the tower kettle, while boron trifluoride -11 is separated from the top of the tower. The process has the characteristics of simple process and simple operation. However, due to the high water content of the boron trifluoride-methyl ether complex purchased from the market, even after dehydration treatment, the water content in the boron trifluoride-methyl ether complex is still as high as 0.01%-0.03%, and the side reactions caused by water A large number of by-products block pipelines, heat exchangers, etc., and severely corrode equipment, making production unable to proceed continuously and stably. |
| Use | is an effective catalyst for many organic reactions with good stability and can be used as a catalyst for alkylation |
| category | flammable articles in contact with water |
| toxicity classification | poisoning |
| acute toxicity | inhalation-guinea pig LCL0: 50 PPM/4 hours |
| explosive hazard characteristics | open flame can explode |
| flammability hazard characteristics | flammable when exposed to water; flammable dimethyl ether gas when exposed to water |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; separate from oxidant and water-containing items |
| fire extinguishing agent | dry powder, dry sand |
| auto-ignition temperature | 455 °F |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
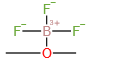
Supplier List
Spot supply
Product Name: Boron trifluoride methyl etherate Visit Supplier Webpage Request for quotationCAS: 353-42-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Boron trifluoride dimethyl etherate Request for quotation
CAS: 353-42-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 353-42-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Boron trifluoride methyl etherate Visit Supplier Webpage Request for quotationCAS: 353-42-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Boron trifluoride dimethyl etherate Request for quotation
CAS: 353-42-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 353-42-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History

